Just as I was about to
board this super cool
all-electric bus
in
Paris
the other day . . .

Calvin Luther Martin, PhD
May 2, 2022
First it was battery-operated electric cars, starting with hybrids (gas & electric), then 100% electric. The batteries in all these vehicles are lithium-ion (Li-ion). (Scroll down the page to see how they work.)
Elon Musk scored big with his Teslas. The only problem is Tesla batteries tend to ignite spontaneously and—oh no!—explode, going into what’s called thermal runaway. Horrific fires that can’t be extinguished by water or chemicals. Click here and here and here.


Electric car batteries look like large wafers—sheets of wafers laid side-by-side, illustrated above.
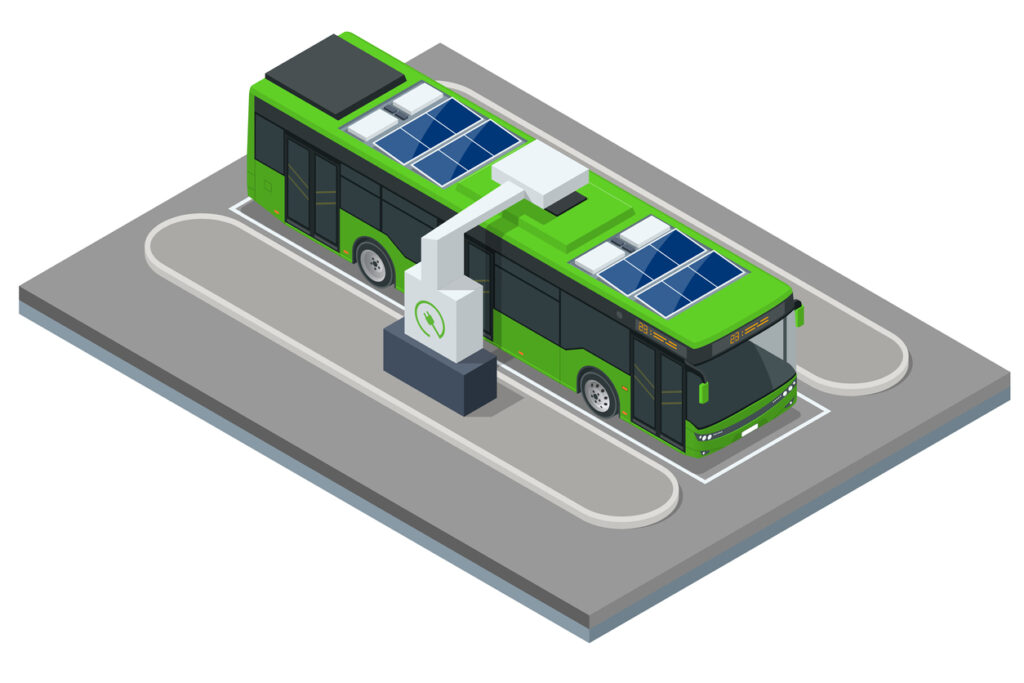
Now the cool thing is all-electric buses. City (municipal) buses, like the one above. Note the solar panels on top: help charge the batteries. This particular bus is being recharged at a charging station.
The lithium-ion batteries are installed in the roof, unlike electric cars. In the image above, the white rectangular boxy structures behind the solar panels appear to be the lithium-ion cells. Some manufacturers, like the French bus manufacturer Bolloré, seem to install them beneath the solar panels.
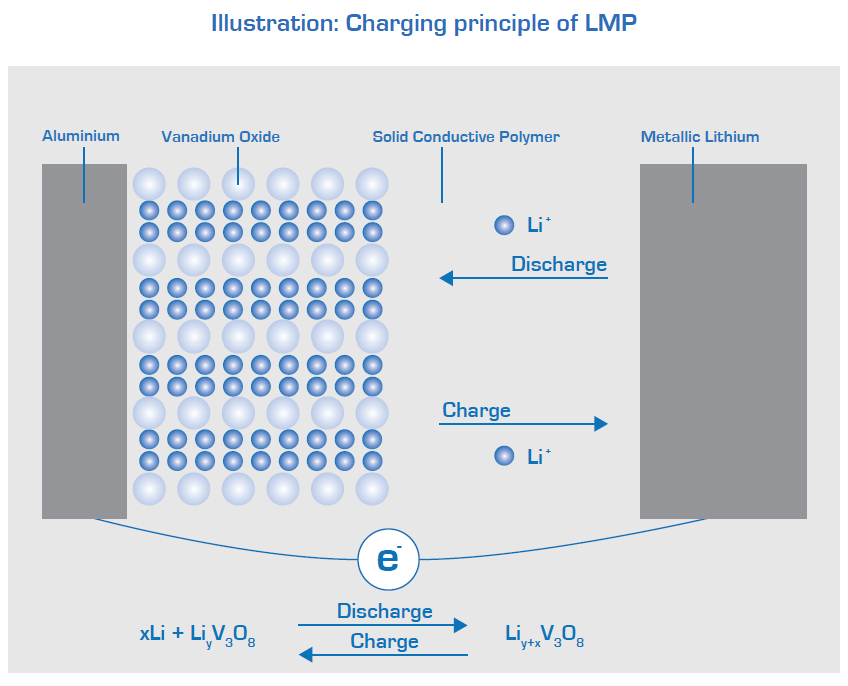
This, above, is a Bolloré Lithium-ion Metal Polymer (LMP) battery, made exclusively and patented by Bolloré.
The text below, from the Bolloré website, explains how Bolloré’s Li-ion battery differs from conventional Li-ion batteries, such as Tesla batteries and those used in solar and wind BESS (Battery Energy Storage Systems).
Bolloré bus batteries, shown above, resemble drawers inserted into a clothes dresser, or case.
Cases of these batteries can be attached to one another in modular fashion, like snapping Legos together .


for this self-congratulation



An electric BlueBus (12m) made by Bolloré caught fire after a battery explosion in Paris.
A video recording shows the start of the fire, which consumed an electric RATP bus on Friday, April 29, 2022. There were no injuries.
The bus burst into flames within seconds of a small explosion on the roof, where the batteries are installed. This was quickly followed by huge flames engulfing the entire body.
The Line 71 bus caught fire in the 13th arrondissement of Paris in the morning, mobilizing around 30 firemen, according to the firefighters contacted by Le Parisien.
It is a 100% electric vehicle, from the Bolloré BlueBus 5SE series, like the bus that burned at the beginning of April .
This afternoon, the RATP decided to temporarily withdraw its fleet of 149 Bolloré electric BlueBuses from service.
Answer: Hydrogen fluoride gas and hydrofluoric acid vapor
Lithium Hexafluorophosphate
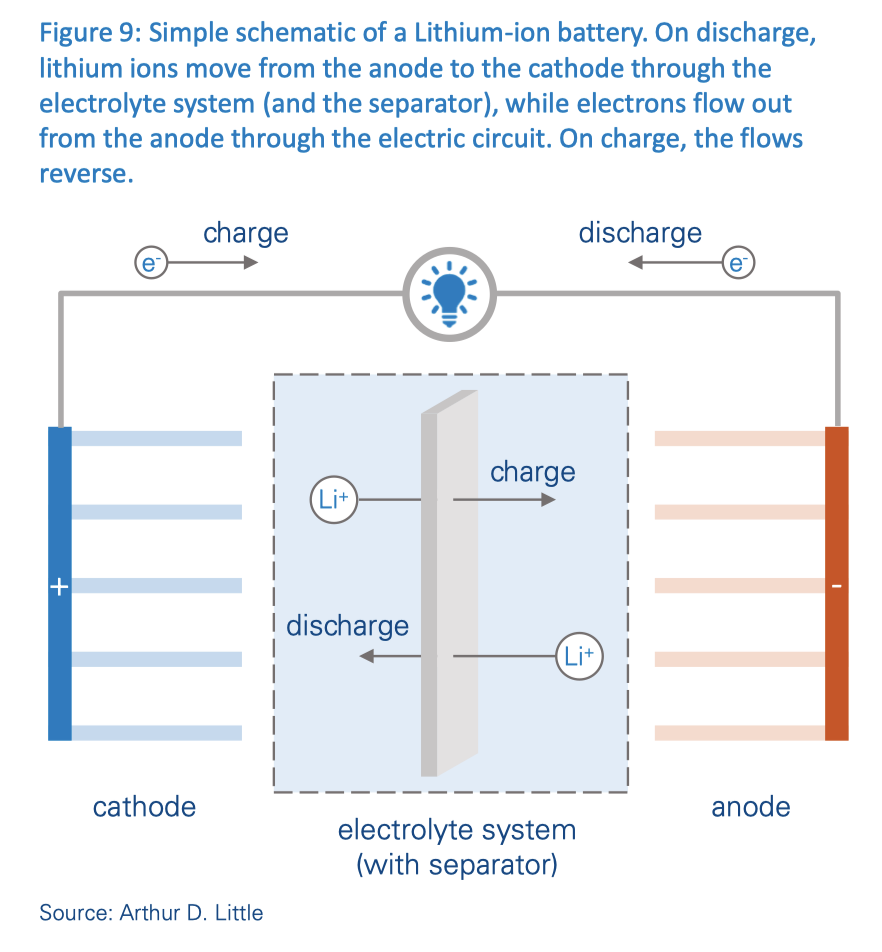
Here is a schematic diagram of a typical lithium-ion battery, as used in solar & wind BESS (Battery Energy Storage Systems).
It’s misleading to call these “Lithium-Ion” batteries. They should really be called “Lithium-Hexafluorophosphate” batteries — to draw attention to the HF gas and acid exposure when these batteries ignite or explode.
The electrolyte in lithium-ion batteries is typically lithium hexafluorophosphate. This is the source of the fluorine in the hydrogen fluoride gas (vapor or mist or smoke) and hydrofluoric acid ejected during thermal runaway or fire or explosion.
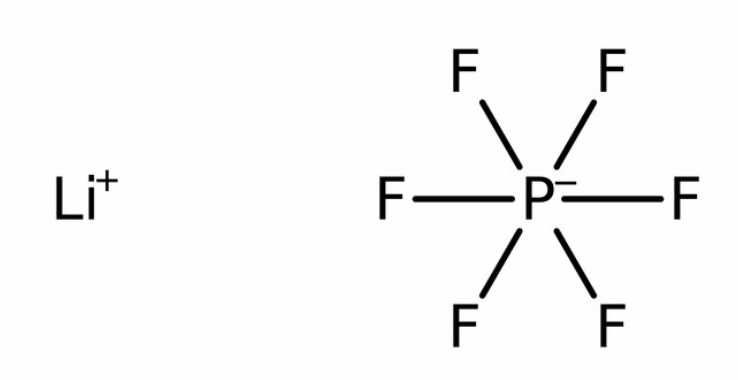
LiPF6

Lithium-ion Metal Polymer (LMP) batteries differ from Li-ion electrolyte batteries in that they use a polymer gel or solid polymer instead of a liquid electrolyte, which, as I noted above, is typically lithium hexafluorophosphate. You can see this illustrated in the diagram. The polymer frequently contains fluoride.
Hence, when LMP batteries explode and burn, as they did last week on the Bolloré bus in Paris, the smoke will contain HF acid.
I recommend the Wikipedia article, below, for further detail.

The imagery (left) is cute and compelling—and deceptive.
There is a horrific side to these electric vehicles. Caveat emptor!






3 thoughts on “Just as I was about to board this super-cool all-electric bus in Paris the other day . . .”
Here’s a news article from FranceTV Info…..Paris RATP temporarily withdraws 149 electric buses from service after 2 vehicles catch fire in less than a month…..
https://www.francetvinfo.fr/economie/transports/paris-la-ratp-retire-temporairement-149-bus-electriques-de-la-circulation-apres-l-incendie-de-deux-vehicules-en-moins-d-un-mois_5109424.html
Not to mention the energy needed to mine for battery materials, the ecological impact of such mining, and the cost/benefit ratio over the long term. The common environmental side effects of lithium mining are water loss, ground destabilization, biodiversity loss, increased salinity of rivers, contaminated soil and toxic waste. Until battery technology improves it seems like a poor (or at the very least, equally poor) substitute for traditional fuel engines…and often charging stations are still powered by power plants which burn fuels. Not to mention the range and recharging problems…
The range of such vehicles (Tesla, for this comparison) is vastly inferior to gasoline engine vehicles which take less than five minutes to refuel, as opposed to upwards of ten hours for a full charge of a Tesla. Even if you had a Tesla “Supercharger” at every stop and you wanted to quick charge you’re only getting about 200 miles in 15 minutes at an official charging station or thirty minutes for a full charge. A trip from Northern NY to Central Florida (for example) would consist of a minimum of 6-7 stops of minimum 15 minutes, or 4-5 stops of over thirty minutes each (IF you find a Supercharge Station along your route where you need them).
Hopefully new technologies will solve this problem in the long term, (https://www.energylivenews.com/2020/09/02/us-startup-unveils-battery-made-from-nuclear-waste-that-could-last-up-to-28000-years/) but it’s hard to say when or if this technology will become viable in the consumer markets…
Shocking!