How safe is your
electric car?
Watch this video
of a cargo ship,
loaded with
electric cars,
go up in smoke!


Calvin Luther Martin, PhD
February 22, 2022

- 1
As I write these words, a cargo ship loaded with Porches, Audis, and Bentleys is adrift and on fire off the Azores—a fire almost certainly caused by one of the cars exploding and going into what’s called thermal runaway.
Why would a vehicle explode? Because of the volatile lithium-ion batteries installed. What follows is a chain reaction.
- 2
The ship is toast. Trying to extinguish the flames with water is pointless: Water doesn’t work on lithium-ion battery fires. (Click here and here to find out why. It’s called thermal runaway in the fire suppression business and it is typical of lithium-ion fires.)
Note that one of the freighters in the video, below, is not the burning ship. I can only guess as to why the film crew included it in the news video.

(Click the X in the upper right corner of this box, "How to enlarge pages," to make it disappear.)
- 3
Tragically, the men (and women?) being lowered by helicoper onto the stricken ship are also toast.
The lithium hexafluorophosphate electrolyte in lithium-ion batteries turns into hydrofluoric gas and acid (HF) when the batteries ignite or explode.

Lithium Hexafluorophosphate
Here is a schematic diagram of a typical lithium-ion battery, as used in solar & wind BESS (Battery Energy Storage Systems).
It’s misleading to call these “Lithium-Ion” batteries. They should really be called “Lithium-Hexafluorophosphate” batteries — to draw attention to the HF gas and acid exposure when these batteries ignite or explode.
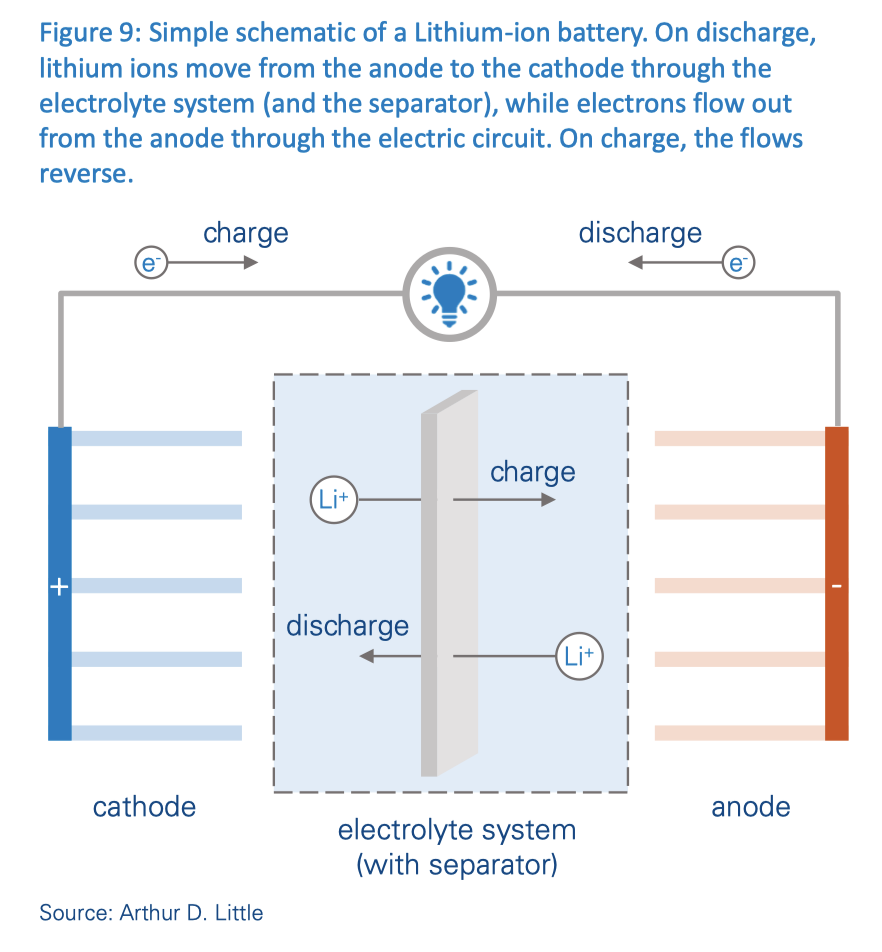
The electrolyte in lithium-ion batteries is typically lithium hexafluorophosphate. This is the source of the fluorine in the hydrogen fluoride gas (vapor or mist or smoke) and hydrofluoric acid ejected during thermal runaway or fire or explosion.
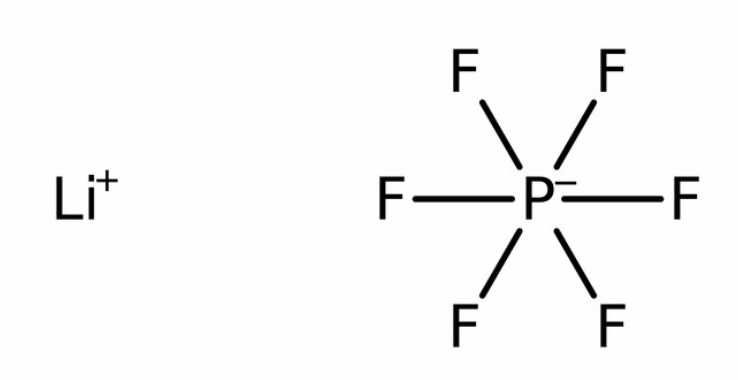
LiPF6


This imagery is cute and compelling—and deceptive.
There is a horrific side to these vehicles.

8 thoughts on “How safe are electric cars?”
Hardly considered is the totally electric powered accessories, which in the case of cars, includes even the door opening mechanisms.
When a Tesla crashes, there have been deaths of occupants who were unable to open the doors or windows to escape the flames of the broken battery.
It is mind boggling to think that such an unsafe condition got past regulators. Oh wait, corporations own the regulators! My bad!
Editor’s response: Click here. From Europe, judging by the license plates and the look of the vehicles. Sent to me by someone in the UK last week.
While the facts and current practical actions of this article are both true and well presented, rather than totally dissing electric cars per se, a plan to a possible solution needs to be presented. After all, fires and destruction of ICE cars do also occasionally occur, albeit not with such deadly results and difficulty to deal with. Let me ask some questions that need to be answered.
1. How frequent are such spontaneous Li-ion battery thermal runaway explosions. (This might provide some measure of risk/benefit analysis.)
2. Since such explosions do not happen very frequently, there must be some specific cause of them and therefore, under what conditions do they occur.
3. Given that sufficient research is done to find out the conditions causing such explosions, is it not possible to change their design and manufacturing so that the chances of such explosions are reduced by several orders of magnitude.
4. Are there not other kinds of batteries that might be used to power electric cars, which would not have this problem?
4. Finally, is it possible to have some warning of when the condition of the battery is such that an explosion may be immanent, and even to have an on board method of preventing such explosion from occuring (even by means of destruction of the further usefulness of the battery, if necessary).
Batteries overheating!
https://www.thejournal.ie/fitbit-recall-google-us-watch-5699301-Mar2022/
Imagine having one of those in your garage? No thanks!
‘Felicity Ace is reportedly carrying around 4,000 vehicles, including Porsches, Audis and Bentleys, estimated to be valued at over $400 million. Some are also believed to be electric vehicles with lithium-ion batteries, which has complicated the firefighting effort.’
https://gcaptain.com/felicity-ace-response-stretches-into-second-week-as-fire-continues-to-burn/
‘Another ongoing ro-ro fire is on a Grimaldi/Euroferry vessel operating between the Greek port of Igoumenitsa to Brindisi, in eastern Italy. Catching fire at 9:30 PM (EST) on 17 February, one crewmember has been sent ashore with injuries, two persons are reported trapped on the car deck, with all other passengers and crew reported as safe. Initial word is that the fire started on the cargo decks and the video below shows extensive damage to trucks stowed on the upper decks.’
‘Grimaldi is no stranger to fires onboard ro-ros with Grande Europa having a fire onboard in the Mediterranean in 2019 and Grande America burning and then sinking in the Bay of Biscay in 2020. After these two events, Grimaldi stated, “…with reference to rolling freight, the Grimaldi Group requests that there be more controls on car batteries, which often cause short-circuits on board vessels, as well as in port terminals.”…’
https://gcaptain.com/electric-vehicles-and-maritime-transportation-fire-hazards-identified/
Seems as if this is the Age of Fixing Things That Aren’t Broken with Things That Make Everything Worse.
Thank you for highlighting this.
On the subject of onshore battery “farms,” as a colleague noted: “How these BESS [Battery Energy Storage Systems] ever get planning approval is astonishing. The government has a Duty of Care to protect its citizens and the environment and, unfortunately, where renewable energy technologies are involved, that is not the case.”
Please see:
https://www.dailymail.co.uk/news/article-9775467/UKs-battery-farms-spark-fears-explosions.html